As you can see from the article, they don't have names yet but it is all over but the shouting (a bit of which I'm doing here) and completing the 7th "period" is tremendous.
One statement from the article really struck me:
'Karol [Paul Karol, chair of the IUPAC's Joint Working Party] says that researchers will continue seeking "the alleged but highly probable 'island of stability' at or near element 120 or perhaps 126," where elements might be found to exist list enough to study their chemistry.'
I haven't heard the phrase "highly probable" associated with the 'island of stability' before so I'm wondered if things have changed since I last read about this or this guy is just flapping his gums.
I looked for an expanded periodic table and found these:
Pretty nice to see everything laid out in fully expanded form. Quite unexpectedly, I also found this:
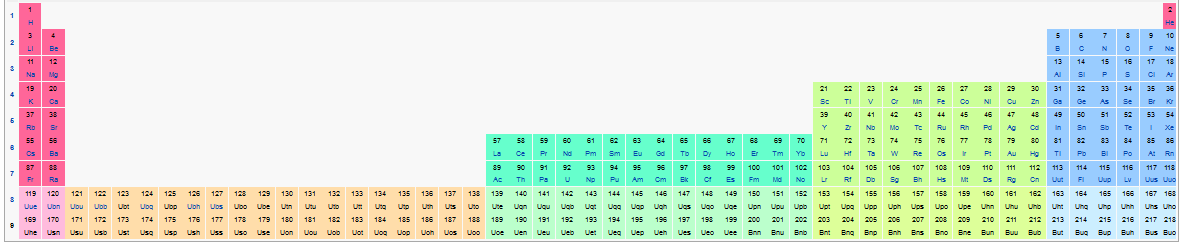
You may have to expand this a bit to see what it is showing but evidently the theory is that that 121 - 138 and 171 -188 are another series of expansion for the table, like the lanthanides (57-70) and actinides (89-102).
Wow, imagine atoms that big being stable with who knows what kind of chemical properties. It boggles the mind. What an exciting time to be alive!
Very Cool Dan.
ReplyDeleteThanks.